European Artificial Intelligence Regulation: Missed Opportunity or Protective Shield

One sector significantly impacted by the use and development of Artificial Intelligence is healthcare and by extension Clinical Research The new Regulation is crucial for AI development as it affects not only technological advancement but also our fundamental rights
Sermes CRO has become the first Contract Research Organization to embrace Farmaindustria’s Code of Conduct for Personal Data Treatment

The company aligns itself with Farmaindustria’s guidelines, which oversee the handling of personal data in the realms of clinical trials and other clinical and pharmacovigilance research
ICH E9 and CDISC: The Master Formula for Excellence in Clinical Research and Data Management

Clinical studies play a key role in the development of new medicinal products and therapies, as well as in evaluating the efficacy and safety of existing medical treatments. To ensure the quality and reliability of the data collected in these studies, it is essential to follow robust guidelines and standards. One of the most important guidelines in this field is ICH E9, published by the International Council on Harmonization (ICH).
Inspiring diversity, transformative inclusion

On the International Day of Persons with Disabilities, December 3, we once again affirm what defines us as an organization: the employment integration of individuals with disabilities, striving for their full integration in terms of employment, social participation, salary, and emotional well-being.
Assessment and approval of clinical trials in CTIS: Theoretical versus actual timelines

Managing clinical trials on the new European clinical trials platform, CTIS, involves taking into account not only the theoretical timelines foreseen in EU CTR 536/2014, but also the actual time it takes for Member States to assess and approve (if applicable) clinical trials submitted via this route.
The CDISC Standards: The Success Formula for Clinical Data
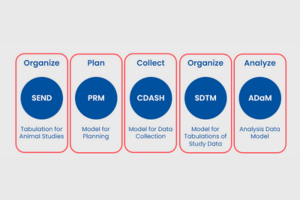
In the world of clinical research, data exchange and analysis play a key role in the development of safe and effective treatments. The Clinical Data Interchange Standards Consortium (CDISC) have proven to be the formula for success in achieving effective harmonization and standardization in clinical data exchange. In this article, we will explore how CDISC standards have revolutionized the way data is presented, analyzed and shared in medical research, improving the quality of clinical trials and accelerating progress towards new therapeutic advances.
Payment of clinical trial fees: news and exemptions

Find out about the five cases that are exempt from paying fees to conduct clinical trials, following legislative changes affecting fees on medicinal products, medical devices, cosmetic products, and personal care products.
Julio Bonis, appointed as Artificial Intelligence & Big Data director at Sermes CRO

Home Services Clinical Trial Regulation: CTIS experts Full Service CRO Advanced Therapy Artificial Intelligence Solutions Customized Technology Solutions Clinical trial support personnel Patient Centric Unit Who we are Blog Careers Contact Our Blog Julio Bonis, appointed as Artificial Intelligence & Big Data director at Sermes CRO We strengthen our Artificial Intelligence & Big Data department […]
Analysis of Express Import Authorizations for medical devices

In exceptional circumstances, healthcare professionals may be faced with the need to use medical devices that have not obtained the CE marking or are to be used under conditions other than those authorized. In this article, we will analyze express authorizations for the import of medical devices, the legislation applicable to them and the documentation required to apply for them.
Bravo for diversity!

In the LGTBI pride week we want to state, once again, what defines us as an organization: at Sermes CRO we are a company with a soul, a company in which the human – and social – component have a main weight. When we say “bravo for diversity” it’s not just words. We are talking about facts. Do you want to know why?